Abstract
Across the tropics, climate change is increasing the frequency and severity of wildfires, exposing tropical forests to the risk of shifting into an open vegetation state. A recent satellite analysis of the Amazon basin suggests this might happen first in floodplains where forests are particularly fragile. We studied floodplain landscapes of the middle Rio Negro, covering ~ 4100 km2 at the Central Amazon region, where forest ecosystems are dominant. We used Landsat images to map 40 years of wildfire history and test the hypothesis that repeatedly burnt forests fail to regenerate and can be replaced by white-sand savanna ecosystems. In the field, using a chronosequence of ‘time after the first fire’, we assessed changes in tree species composition, herbaceous cover and topsoil properties. Here we show that when these forests are repeatedly disturbed by wildfires, their soil gradually loses clay and nutrients and becomes increasingly sandy. In synchrony, native herbaceous cover expands, forest tree species disappear and white-sand savanna tree species become dominant. This drastic ecosystem shift happened within 40 years, likely accelerated by topsoil erosion. When recurrent fires maintain floodplain forests in an open vegetation state, topsoil erosion intensifies, transforming clay-rich soils into white-sand soils that may favour savanna tree species. Our findings reveal that white-sand savannas may expand through seasonally flooded ecosystems at the core of the Amazon, facilitated by wildfires.
Similar content being viewed by others
Highlights
-
We tested whether burnt floodplain forests of the Amazon are recovering or being replaced by white-sand savanna ecosystems.
-
Soil and tree species composition after wildfires shift in concert, causing original forests to change and resemble white-sand savannas within 40 years.
-
Our findings reveal that white-sand savanna ecosystems are expanding at the core of the Amazon.
Introduction
In recent decades, massive wildfires penetrated deep into tropical forests across the Amazon (Aragão and others 2007; Alencar and others 2015; Flores and others 2017a) and Indonesia (Page and others 2002; Lohberger and others 2017), heating debates of whether these forests could persist in a drier future. For thousands of years, savanna expansion over forest has been mostly associated with drier climatic conditions and increased fire occurrence (Mayle and Power 2008). Normally, the closed canopy of tropical forests precludes grass growth, keeping forest flammability low (Charles-Dominique and others 2018; van Nes and others 2018). During drought, however, forests can dry sufficiently for fires to percolate (Aragão and others 2007; Flores and others 2014; Alencar and others 2015), killing trees and opening the understory for flammable herbaceous plants (Cochrane and others 1999; Silvério and others 2013; Brando and others 2014).
It has been hypothesized that under recurrent fire, tropical vegetation can be trapped in an open state with sparse tree cover (Grady and Hoffmann 2012), allowing the expansion of fire-prone vegetation (Hoffmann and others 2012). Yet, evidence for the expansion of native savannas in the tropics remains limited. The best documented cases have been the result of centuries of intensive land use with fire and cattle ranching, leading to soil erosion and the colonization of few savanna tree species, as well as non-native invasive grasses (Borhidi 1988; Cavelier and others 1998; Sansevero and others 2020). Particularly in the Neotropics, the widespread use of African grasses for cattle production has allowed these plants to become invasive among the native vegetation (Williams and Baruch 2000), where they persist as dominant species (Veldman and Putz 2011; Silvério and others 2013; Sansevero and others 2020). As a result, some argue that actually forests are being replaced by degraded ecosystems that largely differ from savannas in terms of plant species composition (Barlow and Peres 2008; Veldman 2016).
In many parts of the tropics, savannas have relatively poorer and sandier soils, compared to forests (Bond 2010; Veldman and Putz 2011; Silva and others 2013; Lehmann and others 2014; Gray and Bond 2015; Paiva and others 2015; Veenendaal and others 2015; Bueno and others 2018). This pattern is common in tropical South America, but it becomes particularly clear in the Amazonian system. Throughout the Amazon basin, islands of white-sand savanna contrast sharply with the clay-rich forests that dominate the landscape (Prance 1996; Adeney and others 2016). The flora and fauna of white-sand ecosystems are also unique, including many endemic species (Ferreira 2009; Capurucho and others 2020; Costa and others 2020), which justifies their reputation as biogeographical islands in Amazonia (Prance 1996). Their origin is often attributed to the long-term sedimentation of ancient fluvial systems (Rossetti and others 2012) and podzolization in water-logged areas, although past fires and indigenous clearing are also plausible mechanisms (Prance and Schubart 1978; Goulding and others 1988; Adeney and others 2016).
White-sand savannas are found across the whole Amazon, covering an area of 87,500 km2, but in the Rio Negro basin they are particularly common (Adeney and others 2016). The Negro River originates in ancient and weathered soils of the Guianan Shield, from where it mainly carries nutrient-poor sand sediments (Latrubesse and Franzinelli 2005; Junk and others 2011). Local people in the region frequently use white-sand savannas as resting sites, or to obtain natural resources, such as turtle eggs (Goulding and others 1988), Couma (sorva) fruits or Leopoldinia piassaba (piassava) fibres. When searching for natural resources, local traditional people occasionally set fire to facilitate their movements (Levis and others 2018). Grass fires in white-sand savannas may spread to surrounding forests; a process that exists for at least 3000 years (Sanford and others 1985).
In the Amazon, recent studies have shown that seasonally flooded forests are particularly vulnerable to wildfires (Flores and others 2014, 2017a; Maracahipes and others 2014; Resende and others 2014), in part because they accumulate fuel in the topsoil (Kauffman and others 1988; dos Santos and Nelson 2013). Floodplain forests often have root mats that protect the top organic soil from water erosion and facilitate soil nutrient capture (Stark and Jordan 1978). Yet during extreme drought events, root mats become flammable, and wildfires can propagate across extensive forest areas, killing up to 90% of all trees (Flores and others 2014, 2016; Resende and others 2014). After fire, recovery is so slow that burnt forests can be easily trapped in an open state by recurrent fires (Flores and others 2016). Studies across varying ecosystems, including tropical forests, have demonstrated that when vegetation cover is lost, topsoil erosion rates increase (Shakesby and Doerr 2006), reducing soil fertility and forest recovery rates (Flores and others 2020). With the loss of soil fertility, environmental filtering could potentially select for plant species that are better adapted to nutrient-poor soils (Bond 2010; Hoffmann and others 2012; Pellegrini and others 2014).
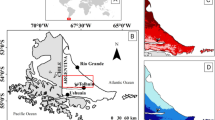
Here we investigate what happens when floodplain forests of the middle Rio Negro are repeatedly burnt by wildfires (Figure 1; Figures S1–S3). Floodplains of this region cover an area of 4100 km2 (Latrubesse and Franzinelli 2005), recently incorporated by the Ramsar Convention into the large Rio Negro site (https://rsis.ramsar.org/ris/2335). Forests dominate the floodplain landscape, but white-sand savannas are also a common scattered vegetation type (Adeney and others 2016). We combined satellite image analysis with field assessments to test the hypothesis that repeatedly burnt forests fail to regenerate and can be replaced by white-sand savanna ecosystems, driven by soil and vegetation changes. Using Landsat imagery, we mapped over 40 years (1973–2014) of forest fire history and produced a chronosequence (space-for-time substitution) of up to 36 years after the first fire. In 21 field sites, including 15 burnt forests, three unburnt forests and three white-sand savannas, we analysed changes in soil properties, herbaceous cover and tree species composition (Table S1, Figure S1, Figure S2). Since the 1970s, total burnt forest area in the study region was under 10,000 ha (Flores et al. 2014). Yet, the mega El Niño of 2015–2016 led to the worse fire season ever recorded, burning 70,000 ha of floodplain forest only around the town of Barcelos (Figure 1), and indicating that drought and wildfire regimes are quickly intensifying in the region.
Floodplain landscapes of the middle Rio Negro, with varying tree cover, and extensive forests burnt in recent decades. The study system covers an area of ~ 4100 km2 (Latrubesse and Franzinelli 2005) and is part of the larger Rio Negro Ramsar site (https://rsis.ramsar.org/ris/2335). Tree cover distribution (MODIS VCF from 2001, DiMiceli and others 2011) is limited to floodable areas (separated by the Wetlands’ mask, Hess and others 2015). (Inner figure) Map of the Amazon Forest showing the study region (red rectangle) embedded in a vast floodplain complex (in gray).
Methods
Study System
We studied floodplain landscapes at the middle Rio Negro, Central Amazonia (Figure 1), where flooding is mostly associated with blackwater rivers, forming ecosystems known as igapó. An annual flood pulse with mean amplitude of 5.5 m inundates the landscape with acid (pH 3.5–4.0) and nutrient-poor water for periods varying from five to 11 months (lowest in January and highest in July; see Table S1) (Junk and others 2011). Despite the low nutrient availability in these floodplains, forests attain high biomass (Saatchi and others 2007). Mean annual rainfall in the region is 2400 mm, with over 100 mm month−1 during the entire year (Agência Nacional de Águas, https://www.ana.gov.br/).
Sampling Design
We studied floodplains within 60 km from Barcelos town, Brazil (0°58S 62°55W) (Figure 1). In this landscape, we identified the dates of 27 fire scars in the forest, of which 13 (48%) had burnt twice (see Fire Detection). Of those 27 scars, we selected 15 for our study based on the number of times they had burnt and the time since their first burn (Figure S1, Table S1), and produced a chronosequence of ‘time after first fire’ ranging from 3 to 36 years. In each independent fire scar, with a unique fire history, we implemented a study plot near the central area. We also used three unburnt mature forests and three white-sand savannas (campinas) as reference sites, well-spread across the study system (Figure S1). White-sand savanna sites were indicated by local informants, who are familiar with their locations commonly hidden within the forest. Among the white-sand savannas available, many were treeless, and therefore, we selected for savanna sites with relatively more trees to allow assessing tree community composition (see Figure S2 a–c). In total, we assessed 21 different sites in the landscape: six burnt once, nine burnt twice, three unburnt forests and three white-sand savannas (Figure S1, Table 1). In each of the 21 sites, we established a 0.6 ha (150 × 40 m) plot, placed at least 100 m far from the nearest river or lake, and at least 30 m far from the fire-scar borders (in the case of burnt sites). The shortest distance between our study sites is 400 m, and they are all separated by navigable rivers. We used long narrow plots to control for topographic variation (dry-phase length) (Table S1).
Fire Detection
To map fire history in our study system, we used Landsat images (MSS, TM and ETM +) from 1973 through 2014. Annual images of this satellite are suitable for detecting understory forest fires, because they reveal fine-scale spectral signs that fade away quickly within two years (Bowman and others 2003; Alencar and others 2015). In Amazonian floodplain forests, where fire impact is severe, Landsat images allow detecting these disturbances particularly well (Flores and others 2014, 2016, 2017a; Resende and others 2014) (see Figure S3). All fire scars used in the study were validated in the field with the presence of local informants, who in many cases knew the fire dates and the person who had ignited them. In others, they had even engaged in trying to prevent the fire from spreading through the forest.
To obtain the year of each fire event, we applied two distinct methods based on Landsat images, one visual and another using time-series analyses. In the visual method, we identified a first fire when spectral patters typical of closed forest changed into open water in images of the high water season and bare soil in images of the low water season (Figure S3). The second fire was identified either when the old fire scar expanded (which we assumed re-burning), or when light green regenerating vegetation returned to bare soil patterns. We then compared the visual fire detection with time-series breakpoint analysis of the normalized difference moisture index (NDMI), derived from Landsat TM 5, 7 and 8 images (Figure S4). NDMI reliably reflects variations in the canopy structure of tropical forests since it saturates less than most common vegetation indices (Dutrieux and others 2016). The NDMI time series were processed for detection of breakpoints, indicative of disturbance and recovery events, using the Breaks For Additive Season and Trend (BFAST) algorithm (Verbesselt and others 2010), processed with the Tucumã Toolbox (Menini and others 2019 at https://github.com/nathmenini/tucuma-toolbox). For each of the 21 study sites, we used a single Landsat pixel to produce the corresponding time series (Figure S4).
Soil Data
In each of the 21 study sites, we collected superficial soil (from 0 to 20 cm including the organic layer) at three evenly spaced positions along the 150 m centre-line of each plot and mixed them to produce one composite sample per site. Collected soils were brought to Manaus to be analysed in the Plant and Soil Laboratory (LTSP) of the National Institute for Amazonian Research (INPA), for mineral fractions (clay, silt and sand), as well as exchangeable cations, available and total (within soil particles) nutrients (as in Quesada and others 2010). pH was extracted in water. Phosphorus (P), potassium (K), iron (Fe), zinc (Zn), manganese (Mn) and copper (Cu) were extracted with Mehlich-1. Calcium (Ca2 +), magnesium (Mg2 +) and aluminium (Al3 +) were extracted with KCl—1 mol/L. Total P, Ca, Al, Mg, K and Na were obtained through soil digestion in concentrated sulphuric acid. Carbon (C) amount and nitrogen stable isotope (15 N) were analysed at the Stable Isotope Facility of University of California (UCDavis), USA.
Statistical Analyses of Soil Data
We selected eight soil variables to study in detail: clay, sand, available P, Ca, K and Mg, C amount and 15 N. These are soil variables with well-known effects on tropical forest dynamics (Quesada and others 2012) and stability (Paiva and others 2015). We analysed changes in those soil variables as a function of ‘time after first fire’. Based on the slopes estimated from these simple relationships, we projected the time in which burnt forest soils would achieve the average levels observed in the white-sand savanna sites. For this, we excluded savanna plots because we had no information on their fire history (Table S1). For these analyses, we used linear and nonlinear (polynomial) models, with the R package ‘ggplot2′, function ‘stat_smooth’ (Wickham 2016), and found that linear models resulted in the best fits.
Using all 20 soil variables, we applied a principal components analysis (PCA) with the R software function ‘prcomp’ (R Core Team 2019), which resulted in two axes that explained 46% (PC1) and 15% (PC2) of the soil variation (Figure S5a). Ranked PCA loadings revealed how much each soil variable was explained by PCA axes (Figure S5b). Clay and sand are shown on both extremes as the soil variables better explained by PC1. We also used Pearson correlation analysis to compare the PC1 with each soil variable (see Figure S6). PC1 correlated well with most soil properties and was considered the ‘soil fertility axis’, separating the more fertile forest soils from the less fertile savanna soils (Figure S5a). PC2 explained variation in soil iron (Fe).
Vegetation Data
In each of the 21 study plots, we measured and identified all trees. Depending on tree size, we considered different sampling widths along the 150 m centre-line of each plot. Small trees (1–10 cm in DBH—diameter at breast height) were included within 2 m of width, medium trees (10—30 cm in DBH) within 20 m, and large trees (> 30 cm in DBH) within 40 m. For all tree species found in each plot, we collected one sample and brought to Manaus. Fertile specimens were deposited at the herbarium of INPA. Using photographs and notes from the field, all tree species were identified at the herbarium of INPA with the help of experts.
We measured herbaceous cover (considering graminoids and herbs above the ground) using the point quadrat method (Sutherland 2006), at six intersections along the 150 m centre-line of each plot (one in every 25 m). Based on the presence or absence at these intersections, we calculated the percentage of herbaceous vegetation cover for each plot. We did not systematically collect and identify the composition of herbaceous plants, but we informally searched for the presence of non-native (exotic) grasses and collected photographs and samples of the dominant species (Figure S2 j-n).
Statistical Analyses of Vegetation Data
We analysed changes in tree composition using the output ordination axes produced by the non-metric multidimensional scaling (NMDS) analysis (Figure S7a). With the R software, ‘vegan’ package (Oksanen and others 2007), function ‘vegdist’, we first produced a dissimilarity matrix with the Bray–Curtis index, using species relative abundance data (relative stem count per species) from the 14 plots that had information on tree species composition (Table S1). We then used the function ‘isoMDS’ from the ‘MASS’ R-package for the NMDS analysis (Venables and Ripley 2002).
From the NMDS analysis (Figure S7a), we found that the second ordination axis (NMDS 2) separated forest and savanna tree communities, whereas the first axis was related to dry phase (or inundation) length (Figure S7b). Using the second ordination axis (NMDS 2), we analysed changes in tree composition as a function of ‘time after first fire’, and based on the estimated slopes, we projected the time needed for the tree community to achieve the average white-sand savanna level along the NMDS 2 axis. Again, we excluded savanna plots from the models because we had no information on their fire history (Table S1). We followed the same procedure with herbaceous cover. To analyse temporal changes in tree composition and herbaceous cover, we used linear functions (which proved more suitable than polynomials) with the ‘ggplot2′ R package (Wickham 2016), as with soil data.
We also explored the existence of tree species indicators of forest and white-sand savanna habitats, using the R package ‘indicspecies’, function ‘multipatt’ (Cáceres and Legendre 2009). Using species abundance data, we estimated each species ‘specificity’ and ‘fidelity’ to their habitat. ‘Specificity’ refers to whether an indicator species is restricted to a habitat or may also occur in other habitats. ‘Fidelity’ refers to whether an indicator species occurs in all sites that belong to their habitat or only a few of the sites.
Dry Phase Length
Dry phase length was estimated for all plots used in the study, except for two forest plots (Table S1), because they were not penetrable by canoe during the high water season. Along the 150 m centre-line of each plot, we used a canoe (6 m long) to collect three well-spaced (by ~ 50 m) measurements of the water column depth, on a known date in the high water season. These depths were compared to the water level of the same day at the Barcelos station, to calculate the average number of days per year that each plot stays dry (above the water level). Historical water level data for Barcelos station (1968–present) were obtained from the Agência Nacional de Águas (http://hidroweb.ana.gov.br/).
Results
Soil Fertility Shift
In unburnt forests, soils had more clay, as well as available and total nutrients compared to white-sand savannas, where soils were sandy and nutrient-poor (Figure 2, Figure S8). However, after the first fire, forest soils started to lose fertility. Despite short increases in the concentration of available P and Ca immediately (3 years) after the first fire, with time, most nutrients and also the clay fraction decreased, while the sand fraction increased, until soils became similar to those found in white-sand savannas (Figure 2). Changes in most soil properties were significant, with the exception of sand (p = 0.1), carbon (p = 0.17) and calcium (p = 0.14). Similar pattern was observed for total soil nutrients, although not statistically significant (Figure S8). As a general pattern, soils continued to change with time despite no further combustion by fire. Based on the time that the eight soil variables take to reach levels observed in white-sand savannas, we estimated an average soil shift within 44 years (Figure 2). If we considered only the sand fraction, this shift would take 131 years.
Changes in soil properties as a function of ‘time after first fire’ in floodable forests. P values refer to linear models (black lines, dashed for p > 0.05 and solid for p < 0.05). White-sand savanna sites were excluded from the analyses and instead shown as mean (± SE) in semi-transparent red colour. Above each plot we show the time in which the mean savanna level is reached. Based on these eight soil variables we estimated an average time of 44 years for the soil shift. Note that changes continue to occur with time despite no further combustion by fire. See Table S1 for site details.
Vegetation Shift
We estimated a significant temporal shift in tree species composition from forest to savanna within 40 years (Figure 3A). This shift was also apparent when comparing sites based on the number of times they were burnt, both when considering all tree species (Figure 4, Table S2), as well as only the forest and savanna indicator species (Table S3). Field data on vegetation structure confirmed our observations that trees in once and twice burnt sites were mostly new recruits that arrived after the fires, and not large remnant trees (Figures S9; S10). After the second fire, tree species that are indicators of white-sand savannas started to appear, such as Byrsonima coniophylla, Maprounea brasiliensis, Myrcia servata and Tachigali goeldiana, while most forest indicator species became rare (Table S3).
Fire triggers a vegetation shift from forest to white-sand savanna. A shift in tree community composition with ‘time after first fire’, indicating that in 40 years, burnt forests become floristically similar to savannas; B simultaneous shift in soil fertility and tree composition, revealing a shift at the ecosystem level; C increase in herbaceous cover with ‘time after first fire’, showing that twice burnt sites persist with open vegetation despite the arrival of trees. In C we added a slight jitter to reveal overlying data points. We show only the sites for which we had both soil and floristic data (Table S1). For (A) and (C), p values refer to linear models (black lines). White-sand savanna sites were excluded from the analysis and instead shown as mean (± SE) in red. D Conceptual model of how forests turn into savannas: fires and flooding maintain low tree cover allowing soils to erode, while trees from white-sand savannas colonize. Central photograph shows the 36-year-old burnt forest B77 (Table S1). Photographs credits to B.M. Flores and Bruce Nelson.
The ‘forest-savanna tree composition axis’ (NMDS 2) was also correlated with the clay fraction (r = −0.75, p = 0.002, Figure 3B), which represented well the overall soil fertility (r = 0.92 with PC1, Figure S5b, Figure S6). Similar patterns were found when all 20 soil variables were plotted against the NMDS 2 (Figure S11). During the same ‘time after first fire’, herbaceous cover increased significantly (p = 0.002, Figure 3C). Moreover, the first ordination axis of tree composition (NMDS 1) was significantly related to ‘dry phase length’ (p = 0.006, Figure S7b).
Discussion
Soil Shift
Our results show that when a first fire destroys the structure of these floodplain forests and a second fire maintains the vegetation open, soil nutrients and finer mineral fractions decrease. On average, soils of burnt forests took only four decades to resemble those of white-sand savannas. This soil shift happens within 20 years for the relatively more mobile nutrients like K and N, within 20–60 years for P, Ca, Mg, C, and clay, and much more slowly for the sand fraction, within 130 years. Nonetheless, all soil properties seem to shift together towards a white-sand savanna state.
Leaching of soluble nutrients and soil erosion are common processes following wildfire in different ecosystems across the world (Shakesby and Doerr 2006; Pellegrini and others 2018; Flores and others 2020). This happens in part because severe fires may increase soil water repellency, leading to higher runoff during rain events (DeBano 2000). Moreover, destruction of the forest litter by fire allows the erosion of finer soil fractions, altering particle size distribution (Certini 2005). Indeed, after the first fire in our study sites, clay decreased and sand increased consistently (Figure 2). Together with clay erosion, total P, Ca, K and Mg concentrations in soil particles decreased as well, although not significantly (Figure S8). Total P is particularly limiting for plants and usually erodes with soil weathering during geological time scales (Lambers and others 2008). Moreover, the 15 N stable isotope is often fractionated by plants, leading to its accumulation in the soil when N is abundant (Martinelli and others 1999). Its depletion in burnt sites indicates increasing N-limitation and that the ecosystem has shifted to a more conservative N cycle. The 15 N has been shown as a proxy for when soils are recovering from disturbance (Davidson and others 2007) or shifting between forest and savanna (Pellegrini and others 2014). Here, we show that it may also indicate a shift from forest to white-sand savanna.
The soil changes triggered by forest fires at the Rio Negro floodplains were fast compared to previous analyses on uplands (Pellegrini and others 2018). Considering that these ecosystems are seasonally flooded by nutrient poor blackwater rivers, the process we observed may be an indication of strong vulnerability to flood erosion (Flores and others 2020). In the forest, trees reduce water currents and turbulence during the high water season, and root mats protect the topsoil, facilitating the retention of fine sediments. Hence, soils are often dominated by clay and silt in these floodplain forests (Latrubesse and Franzinelli 2005). Yet, when wildfires destroy the forest structure and root mats (Flores and others 2016), sediments become vulnerable to erosion. Increased water turbulence in open burnt sites may also allow heavier sand particles to deposit from the Rio Negro waters that carry mostly quartz (Latrubesse and Franzinelli 2005). As a result, the soil of a burnt forest shifts from clayey to sandy. If these sandy soils remain uncovered by forest, the soil shift we observed may be persistent, although studies on palaeoecology and geomorphology are needed to test this hypothesis. Moreover, considering that many Amazonian white-sand savannas are ancient floodplains (Rossetti and others 2012; Adeney and others 2016), our findings may be a hint of how these open ecosystems were formed and maintained by wildfires.
In upland (terra-firme) forests, soil erosion following disturbances may be most severe on hilly terrains (Flores and others 2020). Upland forests usually recover quickly (Poorter and others 2016; Flores and others 2017a), reducing their vulnerability to erosion (Shakesby and Doerr 2006; Flores and others 2020), while promoting soil recovery (Davidson and others 2007). In contrast, Amazonian floodplain forests recover slowly when burnt (Flores and others 2014, 2016, 2017), increasing their vulnerability to erosion.
Vegetation Shift
Fire in these floodplain ecosystems promoted a shift in tree species composition, turning forests into white-sand savannas within 40 years (Figure 3A, Figure S2). Tree community in burnt sites is initially dominated by forest species, yet with time and especially after a second fire event, tree species that normally occur in white-sand savannas become dominant (Figure 4, Table S2), such as Byrsonima coniophylla, Couma utilis, Handroanthus serratifolius, Leptolobium nitens, Myrcia servata, Ouratea discophora, Parkia discolor and Pera bicolor (according to numerous floristic assessments: Ferreira 2009; Vicentini 2004, Damasco and others 2013, Daly and others 2016). Three species in particular: Maprounea brasiliensis, Myrcia servata and Tachigali goeldiana are indicators of white-sand savannas and were abundant in twice burnt sites (Table S3). Other species typical of white-sand savannas, however, had not arrived in burnt sites, such as Couepia bracteosa, Cybianthus fulvopulverulentus, Eugenia florida, Elaeoluma schomburgkiana, Exellodendron coriaceum and Ternstroemia brasiliensis (Ferreira 2009; Vicentini 2004, Garcia-Villacorta and others 2016), possibly due to seed dispersal or recruitment limitations.
The capacity of tropical forests to recover from large and severe wildfires may depend on whether trees can disperse and overcome local environmental filters (Turner and others 1998). Considering that fires killed 100% of the trees and tree seed banks in twice burnt forests, as shown by a previous study (Flores and others 2016), new recruits probably arrived from white-sand savannas nearby (Figure 1A) and germinated in the burnt sites (Figures S9; S10). Tropical forest trees usually rely on animal seed dispersal (Hovestadt and others 1999), an interaction that can be disrupted when animals are sensitive to open habitats (Barlow and others 2006). In contrast, most savanna trees are usually wind dispersed and may be favoured by disturbances (Hovestadt and others 1999). For instance, at the middle Rio Negro (Figure 1), a previous study demonstrated that forest fires persistently change bird communities, with forest bird species disappearing and open habitat bird species becoming dominant (Ritter and others 2012). If birds and other animals from nearby white-sand savannas visit open burnt forests, they might disperse tree seeds and promote the tree compositional shift we observed.
Once trees arrive in burnt sites, they have to face environmental conditions that can potentially limit seedling germination and early growth, such as nutrient-poor soils and high herbaceous cover (Figure 3, Figure S2). Forest tree species are known for being less tolerant to such conditions than savanna tree species (Bond and Midgley 1995; Scholes and Archer 1997; Sankaran and others 2004; Bond 2010; Silva and others 2013; Gray and Bond 2015; Paiva and others 2015). However, with our data, we cannot disentangle the roles of dispersal limitation and environmental filtering in driving ecosystem shifts. Experimental manipulation of seed availability and multiple filters would be the ideal way to unravel the importance of each mechanism.
We also found that herbaceous cover increased in burnt floodplain forests and persisted at levels higher than those of white-sand savannas (Figure 3C). This is a common pattern in disturbed tropical forests, in which fast-growing opportunistic plants expand rapidly benefiting from the flush of nutrients released by fire (Veldman and Putz 2011; Bustamante and others 2012). Although we did not assess herbaceous species composition in the field, we noted that non-native invasive grasses were absent in all sites, probably because of their remoteness. Our study system is far from large-scale deforestation frontiers (Figure 1), where non-native grasses are usually introduced for pasture (Williams and Baruch 2000). Invasive grasses could potentially arrive from small-scale pastures in the study region, yet their seeds would need to travel long distances, crossing wide rivers and forested areas (Figure S1). In burnt sites, we noted instead the dominance of few opportunistic native herbaceous plants (Figure S2). In once burnt sites, a common native species is Scleria secans (Cyperaceae), a climbing herb, well known by locals because it often forms monodominant patches and has razor-like leaves (Figure S2k). In both once and twice burnt sites, we observed the dominance of the annual native herb Hyptis parkieri (Lamiaceae) (Figures S2g and j). The genus Hyptis is known for being fire-adapted and a strong competitor (Veldman and Putz 2011), implying that in burnt sites it may contribute to arrest forest succession.
Our analysis of tree community structure also revealed a significant influence of the dry phase length (Figure S7b). Indeed, tree species distributions in Amazonian floodplains are known for being determined by flood regimes (Wittmann and others 2006, 2010). In these systems, disturbances related to river erosion–deposition processes and the direct effects of inundation on plant physiology are important drivers of vegetation patterns (Salo and others 1986). Our results reveal that fire is another important factor shaping floodplain plant communities.
Abrupt Ecosystem Shift
Forests and savannas can be alternative vegetation states in tropical landscapes, maintained by the interplay of fire, resource availability and plant functional traits (Hirota and others 2011; Staver and others 2011; Hoffmann and others 2012). Our study has provided new insights into how Amazonian islands of white-sand savanna (Prance 1996) may have originated and persisted by wildfires. We have demonstrated that when a first fire destroys forest structure and a second fire traps the vegetation in an open state, soil and tree composition shift in concert. In only four decades, what used to be a forest on clay–silt soil, now resembles a white-sand savanna (Figure 3). If repeated wildfires continue to limit tree recruitment on these sandy and seasonally flooded soils, the shift we observed is likely persistent. Although shifts from forest to savanna are often expected to happen over centuries or millennia (Aleman and others 2019), in Amazonian floodplains, wildfires can push forests into a savanna state rather abruptly, contributing to our understanding of how climate-induced disturbances can cause ecological transitions in the tropics (Holmgren and others 2013; Turner and others 2020).
Although we are not able to infer how soils and plants interact during this ecosystem shift, most likely, the expansion of savannas is accelerated by positive feedback mechanisms. Forests and savannas are often dominated by tree species with contrasting life strategies that allow them to deal with local environmental conditions (Hoffmann and others 2012; Dantas and others 2016). Tree strategies also allow them to reshape environmental conditions, generating positive feedback loops that help stabilize sharp vegetation boundaries (Staal and Flores 2015). For instance, savanna trees typically form thin crowns, allowing grass growth and fire spread (Rossatto and others 2009; Charles-Dominique and others 2018). Their well-developed root systems (Canadell and others 1996; Jackson and others 1996) allow them to resprout after fire and tolerate low resource conditions (Hoffmann and others 2012; Murphy and Bowman 2012; Silva and others 2013; Dantas and others 2016). In contrast, forest trees invest in dense canopies that suppress grass flammability (Rossatto and others 2009; Charles-Dominique and others 2018) and reduce topsoil erosion (Flores and others 2020). Because most forest trees are nutrient demanding (Bond 2010; Silva and others 2013), they benefit from the nutrient-rich litter they produce (Paiva and others 2015).
One major aspect of tropical savannas is their enormous biodiversity, particularly of herbaceous plants that provide critical ecosystem processes (Parr and others 2014; Veldman 2016). White-sand savanna islands of Amazonia are also ancient ecosystems (Cole 1960; Prance 1996; Adeney and others 2016) with endemic biodiversity, and which provide unique services for local societies (Goulding and others 1988; Capurucho and others 2020). Humans have managed those ecosystems for millennia, likely expanding their areas (Prance and Schubart 1978). Hence, differently from other tropical landscapes where savanna-like ecosystems dominated by invasive grasses are expanding over degraded forest (Veldman and Putz 2011; Silvério and others 2013; Sansevero and others 2020), the shift we demonstrate here involves only native species (Figure S2); implying that its consequences must be understood also within the context of traditional landscape management (Levis and others 2018).
El Niño-associated fluctuations in climatic conditions periodically impact tropical forests worldwide (Holmgren and others 2001), causing extreme droughts and wildfires in the Amazon region (Flores and others 2014; Alencar and others 2015). Landscapes of the Rio Negro have witnessed wildfires in the past millennia, yet these events did not threaten forest persistence (Sanford and others 1985). The recent 2015/16 El Niño event, however, led to massive wildfires that burnt an area seven times larger than the whole burnt area accumulated in the previous four decades (Figure 1). Our results indicate that the magnitude and severity of these disturbances will facilitate the spread of white-sand savannas across the core of the Amazon basin, supporting previous evidence (Flores and others 2017a) that floodplains may be an Achilles’ heel of this massive forest system.
Data availability
The data used in this study is part of two large networks, the ATDN (http://atdn.myspecies.info/) and the Synergize (https://synergizeamazonia.wixsite.com/synergize/arboreal-vegetation), and can be used upon request.
References
Adeney JM, Christensen NL, Vicentini A, Cohn-Haft M. 2016. White-sand Ecosystems in Amazonia. Biotropica 48:7–23.
Aleman JC, Blarquez O, Elenga H, Paillard J, Kimpuni V, Itoua G, Issele G, Staver AC. 2019. Palaeo-trajectories of forest savannization in the southern Congo. Biol Lett 15:20190284.
Alencar AA, Brando PM, Asner GP, Putz FE. 2015. Landscape fragmentation, severe drought, and the new Amazon forest fire regime. Ecol Appl 25:1493–1505.
Aragão LE, Malhi Y, Roman-Cuesta RM, Saatchi S, Anderson LO, Shimabukuro YE. 2007. Spatial patterns and fire response of recent Amazonian droughts. Geophys Res Lett 34:L07701.
Barlow J, Peres CA. 2008. Fire-mediated dieback and compositional cascade in an Amazonian forest. Philos Trans Royal Soc London B: Biol Sci 363:1787–1794.
Barlow J, Peres CA, Henriques LM, Stouffer PC, Wunderle JM. 2006. The responses of understorey birds to forest fragmentation, logging and wildfires: an Amazonian synthesis. Biol Conserv 128:182–192.
Bond WJ. 2010. Do nutrient-poor soils inhibit development of forests? A nutrient stock analysis. Plant Soil 334:47–60.
Bond WJ, Midgley JJ. 1995. Kill thy neighbour: an individualistic argument for the evolution of flammability. Oikos 73:79–85.
Borhidi A. 1988. Vegetation dynamics of the savannization process on Cuba. Vegetatio 77:177–183.
Bowman DM, Zhang Y, Walsh A, Williams RJ. 2003. Experimental comparison of four remote sensing techniques to map tropical savanna fire-scars using Landsat-TM imagery. Int J Wildland Fire 12:341–348.
Brando PM, Balch JK, Nepstad DC, Morton DC, Putz FE, Coe MT, Silvério D, Macedo MN, Davidson EA, Nóbrega CC, Alencar A. 2014. Abrupt increases in Amazonian tree mortality due to drought–fire interactions. Proc Natl Acad Sci 111:6347–6352.
Bueno ML, Dexter KG, Pennington RT, Pontara V, Neves DM, Ratter JA, Oliveira-Filho AT. 2018. The environmental triangle of the Cerrado Domain: Ecological factors driving shifts in tree species composition between forests and savannas. J Ecol 106:2109–2120.
Bustamante MM, de Brito DQ, Kozovits AR, Luedemann G, de Mello TR, de Siqueira Pinto A, Munhoz CB, Takahashi FS. 2012. Effects of nutrient additions on plant biomass and diversity of the herbaceous-subshrub layer of a Brazilian savanna (Cerrado). Plant Ecol 213:795–808.
Cáceres M, Legendre P. 2009. Associations between species and groups of sites: indices and statistical inference. Ecology 90:3566–3574.
Canadell J, Jackson RB, Ehleringer JB, Mooney HA, Sala OE, Schulze ED. 1996. Maximum rooting depth of vegetation types at the global scale. Oecologia 108:583–595.
Capurucho JM, Borges SH, Cornelius C, Vicentini A, Prata EM, Costa FM, Campos P, Sawakuchi AO, Rodrigues F, Zular A, Aleixo A. 2020. Patterns and processes of diversification in Amazonian white-sand ecosystems: insights from birds and plants. In: Rull V, Carnaval A, Eds. Neotropical Diversification: Patterns and Processes, . Springer Nature Switzerland AG: Cham, Switzerland. pp 245–270.
Cavelier J, Aide T, Santos C, Eusse A, Dupuy J. 1998. The savannization of moist forests in the Sierra Nevada de Santa Marta, Colombia. J Biogeogr 25:901–912.
Certini G. 2005. Effects of fire on properties of forest soils: a review. Oecologia 143:1–10.
Charles-Dominique T, Midgley GF, Tomlinson KW, Bond WJ. 2018. Steal the light: shade vs fire adapted vegetation in forest–savanna mosaics. New Phytologist 218:1419–1429.
Cochrane MA, Alencar A, Schulze MD, Souza CM, Nepstad DC, Lefebvre P, Davidson EA. 1999. Positive feedbacks in the fire dynamic of closed canopy tropical forests. Science 284:1832–1835.
Coetsee C, Bond WJ, February EC. 2010. Frequent fire affects soil nitrogen and carbon in an African savanna by changing woody cover. Oecologia 162:1027–1034.
Cole MM. 1960. Cerrado, caatinga and pantanal: the distribution and origin of the savanna vegetation of Brazil. Geogr J 126:168–179.
Costa FM, Terra-Araujo MH, Zartman CE, Cornelius C, Carvalho FA, Hopkins MJ, Viana PL, Prata EM, Vicentini A. 2020. Islands in a green ocean: Spatially structured endemism in Amazonian white-sand vegetation. Biotropica 52:34–45.
Daly DC, Silveira M, Medeiros H, Castro W, Obermüller FA. 2016. The White-sand Vegetation of Acre, Brazil. Biotropica 48:81–89.
Damasco G, Vicentini A, Castilho CV, Pimentel TP, Nascimento HE. 2013. Disentangling the role of edaphic variability, flooding regime and topography of Amazonian white-sand vegetation. J Veg Sci 24:384–394.
Dantas VD, Hirota M, Oliveira RS, Pausas JG. 2016. Disturbance maintains alternative biome states. Ecol Lett 19:12–19.
Davidson EA, de Carvalho CJ, Figueira AM, Ishida FY, Ometto JP, Nardoto GB, Sabá RT, Hayashi SN, Leal EC, Vieira IC, Martinelli LA. 2007. Recuperation of nitrogen cycling in Amazonian forests following agricultural abandonment. Nature 447:995–998.
de Resende AF, Nelson BW, Flores BM, de Almeida DR. 2014. Fire Damage in Seasonally Flooded and Upland Forests of the Central Amazon. Biotropica 46:643–646.
DeBano LF. 2000. The role of fire and soil heating on water repellency in wildland environments: a review. J Hydrol 231:195–206.
DiMiceli CM, Carroll ML, Sohlberg RA, Huang C, Hansen MC, Townshend JRG. 2011. Annual Global Automated MODIS Vegetation Continuous Fields (MOD44B) at 250 m Spatial Resolution for Data Years Beginning Day 65, 2000–2010, Collection 5 Percent Tree Cover. College Park, MD, USA: University of Maryland.
dos Santos AR, Nelson BW. 2013. Leaf decomposition and fine fuels in floodplain forests of the Rio Negro in the Brazilian Amazon. J Trop Ecol 29:455–458.
Dutrieux LP, Jakovac CC, Latifah SH, Kooistra L. 2016. Reconstructing land use history from Landsat timeseries: Case study of a swidden agriculture system in Brazil. Int J Appl Earth Observ Geoinform 47:112–124.
Ferreira CAC. (2009). Analise comparativa de vegetacão lenhosa do ecossistema campina na Amazônia brasileira. Ph.D. Dissertation, National Institute for Amazonian Research INPA, Manaus, Brazil.
Flores BM, Piedade MTF, Nelson BW. 2014. Fire disturbance in Amazonian blackwater floodplain forests. Plant Ecol Divers 7:319–327.
Flores BM, Fagoaga R, Nelson BW, Holmgren M. 2016. Repeated fires trap Amazonian blackwater floodplains in an open vegetation state. J Appl Ecol 53:1597–1603.
Flores BM, Holmgren M, Xu C, van Nes EH, Jakovac CC, Mesquita RC, Scheffer M. 2017. Floodplains as an Achilles’ heel of Amazonian forest resilience. Proc Natl Acad Sci 114:4442–4446.
Flores BM, Staal A, Jakovac CC, Hirota M, Holmgren M, Oliveira RS. 2020. Soil erosion as a resilience drain in disturbed tropical forests. Plant Soil 450:11–27.
García-Villacorta R, Dexter KG, Pennington T. 2016. Amazonian white-sand-forests show strong floristic links with surrounding oligotrophic habitats and the Guiana Shield. Biotropica 48:47–57.
Goulding M, Carvalho ML, Ferreira EG. 1988. Rio Negro, rich life in poor water. Amazonian diversity and foodchain ecology as seen through fish communities: SPB Academic Publishing. the University of Michigan. p 200.
Grady JM, Hoffmann WA. 2012. Caught in a fire trap: recurring fire creates stable size equilibria in woody resprouters. Ecology 93:2052–2060.
Gray EF, Bond WJ. 2015. Soil nutrients in an African forest/savanna mosaic: Drivers or driven? S Afr J Bot 101:66–72.
Hess LL, Melack JM, Affonso AG, Barbosa C, Gastil-Buhl M, Novo EM. 2015. Wetlands of the lowland Amazon basin: Extent, vegetative cover, and dual-season inundated area as mapped with JERS-1 Synthetic Aperture Radar. Wetlands 35:745–756.
Hirota M, Holmgren M, Van Nes EH, Scheffer M. 2011. Global resilience of tropical forest and savanna to critical transitions. Science 334:232–235.
Hoffmann WA, Geiger EL, Gotsch SG, Rossatto DR, Silva LC, Lau OL, Haridasan M, Franco AC. 2012. Ecological thresholds at the savanna-forest boundary: how plant traits, resources and fire govern the distribution of tropical biomes. Ecol Lett 15:759–768.
Holmgren M, Scheffer M, Ezcurra E, Gutiérrez JR, Mohren GM. 2001. El Niño effects on the dynamics of terrestrial ecosystems. Trends Ecol Evol 16:89–94.
Holmgren M, Hirota M, Van Nes EH, Scheffer M. 2013. Effects of interannual climate variability on tropical tree cover. Nat Clim Change 3:755–758.
Hovestadt T, Yao P, Linsenmair KE. 1999. Seed dispersal mechanisms and the vegetation of forest islands in a West African forest-savanna mosaic (Comoé National Park, Ivory Coast). Plant Ecol 144:1–25.
Jackson RB, Canadell J, Ehleringer JR, Mooney HA, Sala OE, Schulze ED. 1996. A global analysis of root distributions for terrestrial biomes. Oecologia 108:389–411.
Junk WJ, Piedade MT, Schöngart J, Cohn-Haft M, Adeney JM, Wittmann F. 2011. A classification of major naturally-occurring Amazonian lowland wetlands. Wetlands 31:623–640.
Kauffman JB, Uhl C, Cummings DL. 1988. Fire in the Venezuelan Amazon 1: fuel biomass and fire chemistry in the evergreen rainforest of Venezuela. Oikos 1:167–175.
Lambers H, Raven JA, Shaver GR, Smith SE. 2008. Plant nutrient-acquisition strategies change with soil age. Trends Ecol Evol 23:95–103.
Latrubesse EM, Franzinelli E. 2005. The late Quaternary evolution of the Negro River, Amazon, Brazil: implications for island and floodplain formation in large anabranching tropical systems. Geomorphology 70:372–397.
Lehmann CE, Anderson TM, Sankaran M, Higgins SI, Archibald S, Hoffmann WA, Hanan NP, Williams RJ, Fensham RJ, Felfili J, Hutley LB. 2014. Savanna vegetation-fire-climate relationships differ among continents. Science 343:548–552.
Levis C, Flores BM, Moreira PA, Luize BG, Alves RP, Franco-Moraes J, Lins J, Konings E, Peña-Claros M, Bongers F, Costa FR. 2018. How people domesticated Amazonian forests. Front Ecol Evol 5:171.
Lohberger S, Stängel M, Atwood EC, Siegert F. 2017. Spatial evaluation of Indonesia’s 2015 fire affected area and estimated carbon emissions using Sentinel-1. Global Change Biol 24:644–654.
Maracahipes L, Marimon BS, Lenza E, Marimon-Junior BH, de Oliveira EA, Mews HA, Gomes L, Feldpausch TR. 2014. Post-fire dynamics of woody vegetation in seasonally flooded forests (impucas) in the Cerrado-Amazonian Forest transition zone. Flora-Morphol, Distrib, Funct Ecol Plants 209:260–270.
Martinelli LA, Piccolo MC, Townsend AR, Vitousek PM, Cuevas E, McDowell W, Robertson GP, Santos OC, Treseder K. 1999. Nitrogen stable isotopic composition of leaves and soil: tropical versus temperate forests. Biogeochemistry 46:45–65.
Mayle FE, Power MJ. 2008. Impact of a drier Early–Mid-Holocene climate upon Amazonian forests. Philos Trans Royal Soc London B: Biol Sci 363:1829–1838.
Menini N, Almeida AE, Lamparelli R, Le Maire G, Oliveira RS, Verbesselt J, Hirota M, Torres RD. 2019. Tucumã: A toolbox for spatiotemporal remote sensing image analysis [Software and Data Sets]. IEEE Geosci Remote Sens Mag 7:110–122.
Murphy BP, Bowman DM. 2012. What controls the distribution of tropical forest and savanna? Ecol Lett 15:748–758.
Oksanen J, Kindt R, Legendre P, O’Hara B, Simpson GL, Solymos P, Stevens MH, Wagner H. 2007. Vegan Pack. Commun Ecol Pack 10:631–637.
Page SE, Siegert F, Rieley JO, Boehm HD, Jaya A, Limin S. 2002. The amount of carbon released from peat and forest fires in Indonesia during 1997. Nature 420:61–65.
Paiva AO, Silva LCR, Haridasan M. 2015. Productivity-efficiency tradeoffs in tropical gallery forest-savanna transitions: linking plant and soil processes through litter input and composition. Plant Ecol 216:775–787.
Parolin PD, De Simone O, Haase K, Waldhoff D, Rottenberger S, Kuhn U, Kesselmeier J, Kleiss B, Schmidt W, Pledade MT, Junk WJ. 2004. Central Amazonian floodplain forests: tree adaptations in a pulsing system. Bot Rev 70:357–380.
Parr CL, Lehmann CE, Bond WJ, Hoffmann WA, Andersen AN. 2014. Tropical grassy biomes: misunderstood, neglected, and under threat. Trends Ecol Evol 29:205–213.
Pellegrini AFA, Hoffmann WA, Franco AC. 2014. Carbon accumulation and nitrogen pool recovery during transitions from savanna to forest in central Brazil. Ecology 95:342–352.
Pellegrini AF, Ahlström A, Hobbie SE, Reich PB, Nieradzik LP, Staver AC, Scharenbroch BC, Jumpponen A, Anderegg WR, Randerson JT, Jackson RB. 2018. Fire frequency drives decadal changes in soil carbon and nitrogen and ecosystem productivity. Nature 553:194–198.
Poorter L, Bongers F, Aide TM, Zambrano AM, Balvanera P, Becknell JM, et al. 2016. Biomass resilience of Neotropical secondary forests. Nature 530:211–214.
Prance GT. 1996. Islands in Amazonia. Philos Trans Royal Soc of London B: Biol Sci 351:823–833.
Prance GT, Schubart HO. 1978. Notes on the vegetation of Amazonia I. A preliminary note on the origin of the open white sand campinas of the lower Rio Negro. Brittonia 30:60–63.
Quesada CA, Lloyd J, Schwarz M, Patino S, Baker TR, Czimczik C, Fyllas NM, Martinelli L, Nardoto GB, Schmerler J, Santos AJ. 2010. Variations in chemical and physical properties of Amazon forest soils in relation to their genesis. Biogeosciences 7:1515–1541.
Quesada CA, Phillips OL, Schwarz M, Czimczik CI, Baker TR, Patiño S, et al. 2012. Basin-wide variations in Amazon forest structure and function are mediated by both soils and climate. Biogeosciences 9:2203–2246.
R Core Team. (2019). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/.
Ritter CD, Andretti CB, Nelson BW. 2012. Impact of past forest fires on bird populations in flooded forests of the Cuini River in the Lowland Amazon. Biotropica 44:449–453.
Rossatto DR, Hoffmann WA, Franco AC. 2009. Differences in growth patterns between co-occurring forest and savanna trees affect the forest–savanna boundary. Funct Ecol 23:689–698.
Rossetti DF, Bertani TC, Zani H, Cremon EH, Hayakawa EH. 2012. Late Quaternary sedimentary dynamics in Western Amazonia: Implications for the origin of open vegetation/forest contrasts. Geomorphology 177:74–92.
Saatchi SS, Houghton RA, Dos Santos Alvala RC, Soares JV, Yu Y. 2007. Distribution of aboveground live biomass in the Amazon basin. Global Change Biol 13:816–837.
Salo J, Kalliola R, Häkkinen I, Mäkinen Y, Niemelä P, Puhakka M, Coley PD. 1986. River dynamics and the diversity of Amazon lowland forest. Nature 322:254–258.
Sanford RL, Saldarriaga J, Clark KE, Uhl C, Herrera R. 1985. Amazon rain-forest fires. Science 227:53–55.
Sankaran M, Ratnam J, Hanan NP. 2004. Tree–grass coexistence in savannas revisited–insights from an examination of assumptions and mechanisms invoked in existing models. Ecol Lett 7:480–490.
Sansevero JB, Garbin ML, Sánchez-Tapia A, Valladares F, Scarano FR. 2020. Fire drives abandoned pastures to a savanna-like state in the Brazilian Atlantic Forest. Perspect Ecol Conserv 18:31–36.
Scholes RJ, Archer SR. 1997. Tree-grass interactions in savannas. Annu Rev Ecol Syst 28:517–544.
Shakesby RA, Doerr SH. 2006. Wildfire as a hydrological and geomorphological agent. Earth-Sci Rev 74:269–307.
Silva LC, Hoffmann WA, Rossatto DR, Haridasan M, Franco AC, Horwath WR. 2013. Can savannas become forests? A coupled analysis of nutrient stocks and fire thresholds in central Brazil. Plant Soil 373:829–842.
Silvério DV, Brando PM, Balch JK, Putz FE, Nepstad DC, Oliveira-Santos C, Bustamante MMC. 2013. Testing the Amazon savannization hypothesis: fire effects on invasion of a neotropical forest by native cerrado and exotic pasture grasses. Philos Trans Royal Soc London B: Biol Sci 368:20120427.
Staal A, Flores BM. 2015. Sharp ecotones spark sharp ideas: comment on ‘Structural, physiognomic and above-ground biomass variation in savanna–forest transition zones on three continents–how different are co-occurring savanna and forest formations?’ by Veenendaal and others (2015). Biogeosciences 12:5563–5566.
Stark NM, Jordan CF. 1978. Nutrient retention by the root mat of an Amazonian rain forest. Ecology 59:434–437.
Staver AC, Archibald S, Levin SA. 2011. The global extent and determinants of savanna and forest as alternative biome states. Science 334:230–232.
Sutherland WJ. (Ed.). (2006). Ecological census techniques: a handbook. Cambridge University Press. p. 194.
Turner MG, Baker WL, Peterson CJ, Peet RK. 1998. Factors influencing succession: lessons from large, infrequent natural disturbances. Ecosystems 1:511–523.
Turner MG, Calder WJ, Cumming GS, Hughes TP, Jentsch A, LaDeau SL, et al. 2020. Climate change, ecosystems and abrupt change: science priorities. Philos Trans Royal Soc London B: Biol Sci 375:20190105.
van Nes EH, Staal A, Hantson S, Holmgren M, Pueyo S, Bernardi RE, Flores BM, Xu C, Scheffer M. 2018. Fire Forbids Fifty-Fifty For. PloS One 13:e0191027.
Veenendaal EM, Torello-Raventos M, Feldpausch TR, Domingues TF, Ceca G, Sykora KV. 2015. Structural, physiognomic and above-ground biomass variation in savanna-forest transition zones on three continents-how different are co-occurring savanna and forest formations? Biogeosciences 12:2927–2951.
Veldman JW. 2016. Clarifying the confusion: old-growth savannahs and tropical ecosystem degradation. Philos Trans Royal Soc B: Biol Sci 371:20150306.
Veldman JW, Putz FE. 2011. Grass-dominated vegetation, not species-diverse natural savanna, replaces degraded tropical forests on the southern edge of the Amazon Basin. Biol Conserv 144:1419–1429.
Venables WN, Ripley BD. 2002. Modern Applied Statistics with S, 4th edn. New York: Springer ISBN 0-387-95457-0.
Verbesselt J, Hyndman R, Newnham G, Culvenor D. 2010. Detecting trend and seasonal changes in satellite image time series. Remote Sens Environ 114:106–115.
Vicentini A. 2004. A vegetação ao longo de um gradiente edáfico no Parque Nacional do Jaú. In: Iwanaga S, Durigan CC, Pinheiro MR, Eds. Janelas para a biodiversidade no Parque Nacional do Jaú: uma estratégia par ao estudo da biodiversidade na Amazônia (SH Borges, . Manaus: Fundação Vitória Amazônica/WWF/IBAMA. pp 117–143.
Wickham H. (2016). ggplot2: Elegant Graphics for Data Analysis. Springer-Verlag New York. ISBN 978–3–319–24277–4, https://ggplot2.tidyverse.org.
Williams DG, Baruch Z. 2000. African grass invasion in the Americas: ecosystem consequences and the role of ecophysiology. Biol Invasions 2:123–140.
Wittmann F, Schöngart J, Montero JC, Motzer T, Junk WJ, Piedade MT, Queiroz HL, Worbes M. 2006. Tree species composition and diversity gradients in white-water forests across the Amazon Basin. J Biogeogr 33:1334–1347.
Wittmann F, Schöngart J, Junk WJ. (2010). Phytogeography, species diversity, community structure and dynamics of central Amazonian floodplain forests. In: Amazonian Floodplain Forests: Ecophysiology, Biodiversity and Sustainable Management (eds Junk WJ, Piedade MTF, Wittmann F, Schongart J, Parolin P), pp. 347–388, Ecological Studies 210. Springer Science & Business Media, Netherlands, Dordrecht.
Acknowledgements
We thank Carolina Levis for numerous useful suggestions, Sally Archibald, José L. Attayde, Raquel Fagoaga, Carlos Fonseca, Marina Hirota, Bruce Nelson, Rafael Oliveira, Carlos A. Quesada, Marten Scheffer, Arie Staal, Joseph Veldman and Eduardo Venticinque for exciting discussions, José Ramos and Alexandro Santos (INPA-Herbarium) for helping identifying all tree species, Layon Demarchi and Aline Lopes for helping identifying herbaceous plants, R. Fagoaga and C. Levis also for field assistance, Erison Gomes and C. A. Quesada (LTSP-INPA) for soil analyses. We thank FAPEAM (Amazonas Research Support Foundation) and CNPq (Brazilian National Research Council) for financial support. IDAM-AM provided logistical support. B.M. Flores was supported by Wageningen University, Coordination for the Improvement of Higher Education Personnel (CAPES) and by São Paulo Research Foundation (FAPESP)—grant #2016/25086-3.
Author information
Authors and Affiliations
Corresponding authors
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Flores, B.M., Holmgren, M. White-Sand Savannas Expand at the Core of the Amazon After Forest Wildfires. Ecosystems 24, 1624–1637 (2021). https://doi.org/10.1007/s10021-021-00607-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10021-021-00607-x